There are many issues that need addressing when delivering life saving medications by way of an injection. The problem starts with the process of readying the syringe that will deliver the medication. The first step should be to completely empty all of the medication from its vial safely including medication that hides in the recesses of its rubber stopper. And another is to deliver all of that medication into a person using the sharpest non manipulated steel needle all while being a syringe that limits its dead space that saves valuable medication from being wasted.
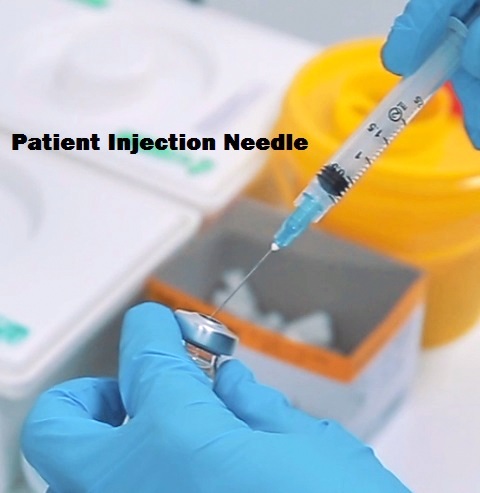
To properly remove medication from a vial a patient injection needle should not be used, though this is done frequently. By using the patient injection needle to draw medication, the tip of the needle microscopically bends causing pain on injection as well as removing strategically placed lubrication which is applied during the manufacturing process to insure a more painless insertion.
Another matter is the risk of a coring event which may shave off rubber stopper particles that may be injected into the patient after the medication is drawn down into the syringe.

Additionally, the needles’ insufficient design does not ensure that the vial is completely empty once the medication is drawn up into the syringe.
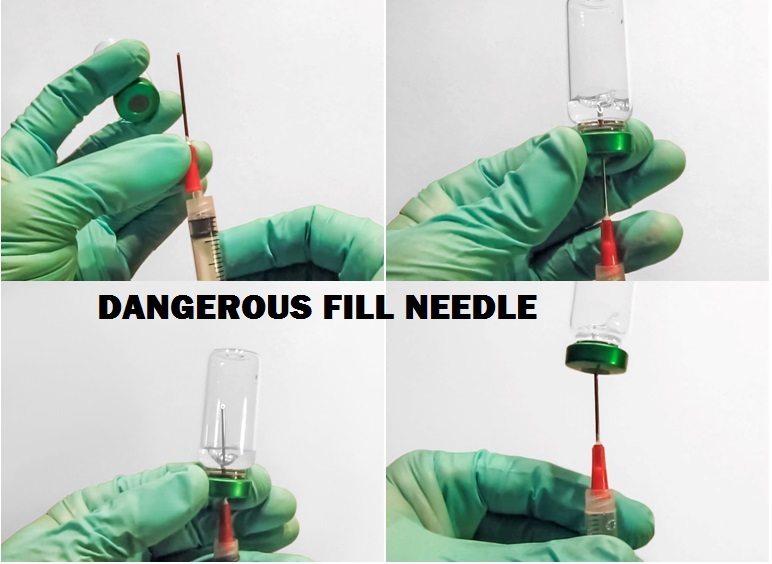
Finally, the use of detachable steel fill needles are dangerous and may contribute to needle stick injuries to both medical personnel and patients.
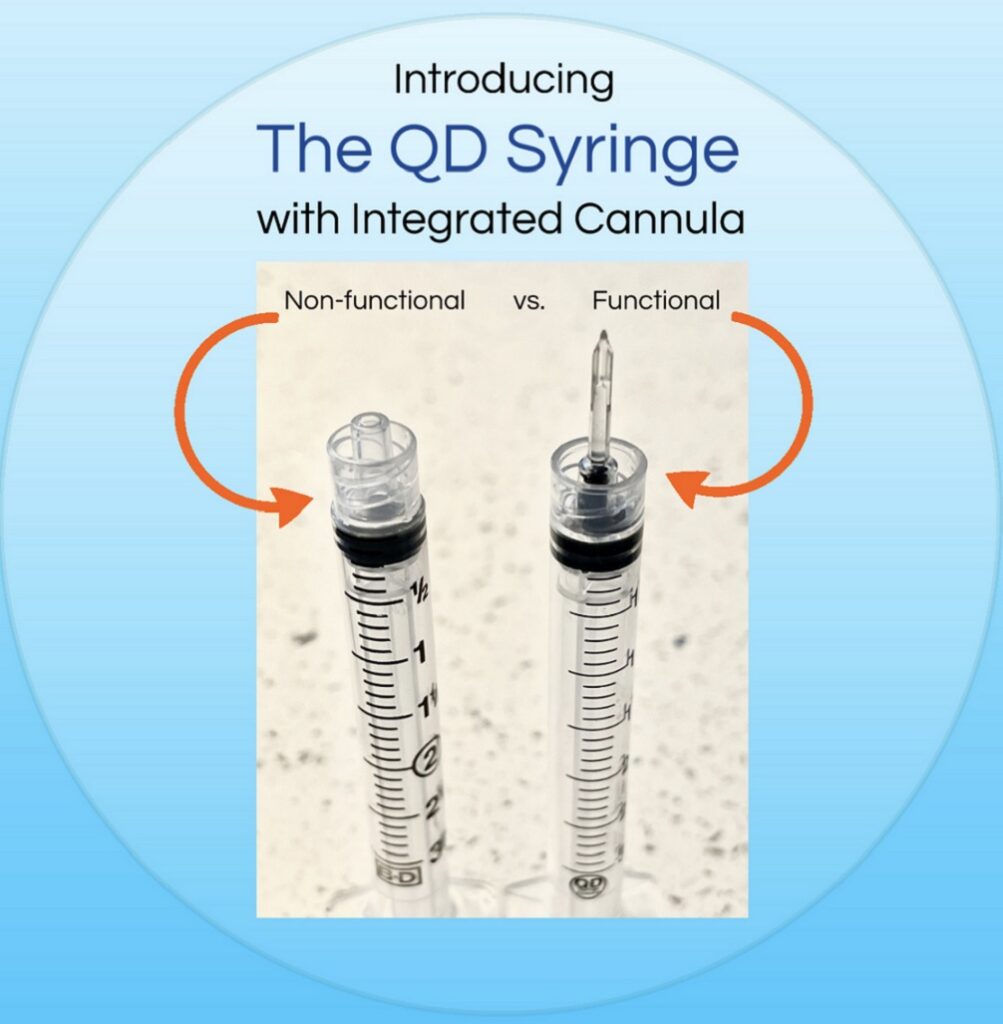
The most effective way is to use a syringe with an integrated cannula as a one-piece-unit: a basic syringe that is functional.

A functional one piece syringe with an incorporated draw down cannula that is non-coring; that is an anti-needle stick cannula,
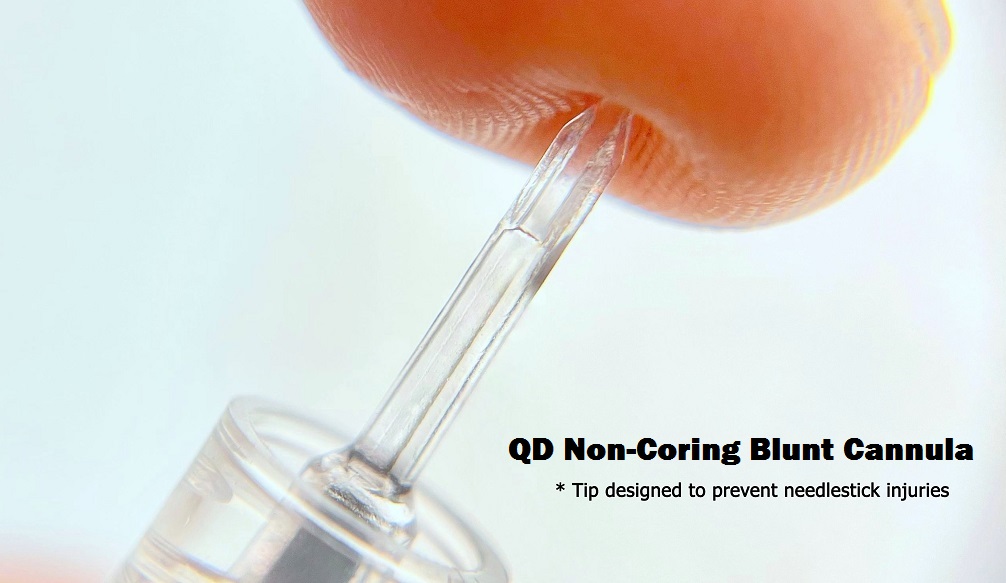
syringe which completes the transfer of medication from a vial into a syringe with strategically placed bilateral channels;
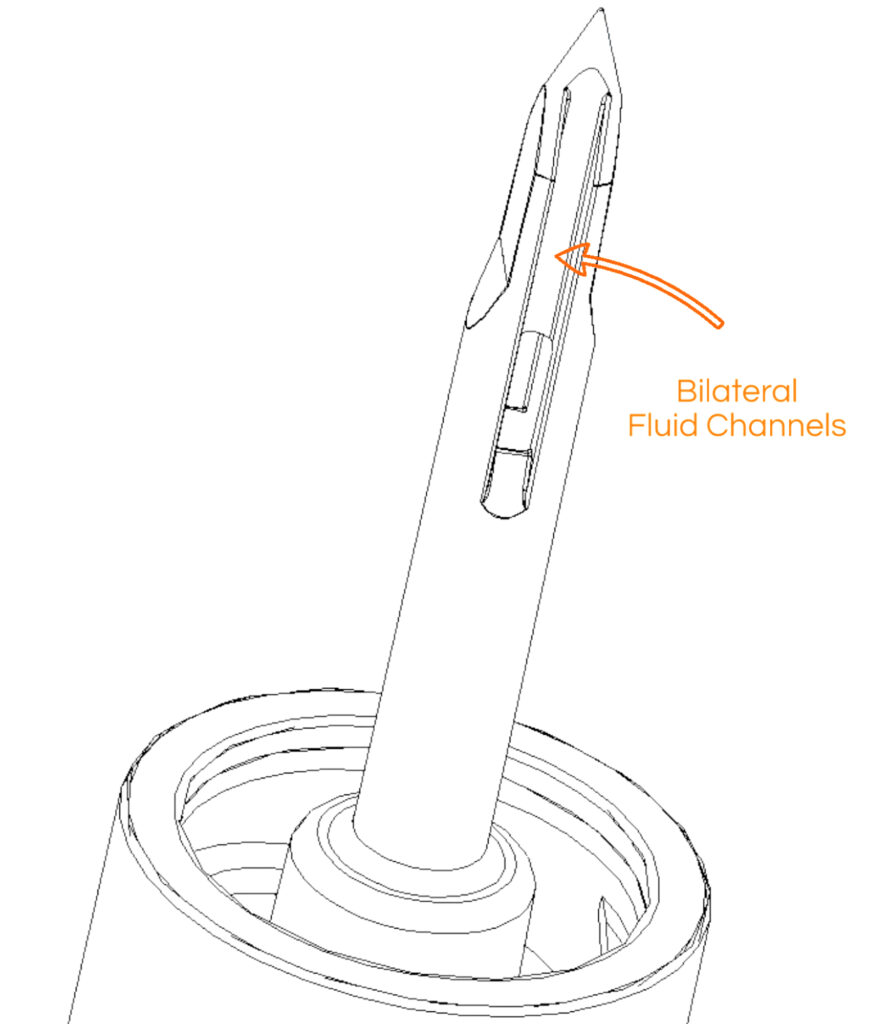
that ensures that once all of the medication within a vial is removed

the final step of patient delivery is attaching its very own injection hub with needle, which has never been used on anything but the patient first; and finally, a syringe that can deliver medications leaving a low residual volume.
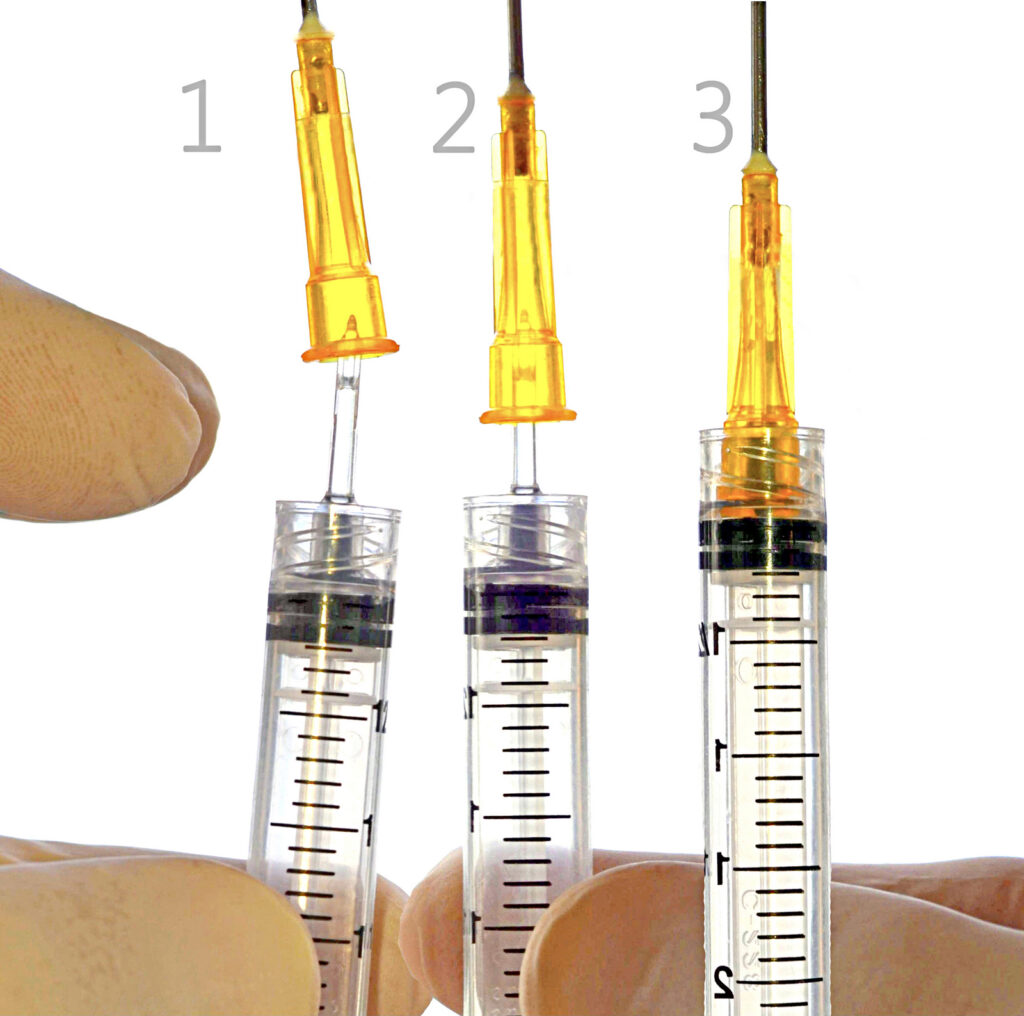
The syringe being described with all of these key attributes is the QD Syringe. The QD Syringe is the next generation low dead space disposable syringes with functional draw capability and detachable needles. The patented design of the QD Syringe includes several innovative concepts that help make it one of the most versatile syringe products available. The QD Syringe consists of an integrated patented draw down tip designed as a ready to use syringe with its unique bilateral fluid flow channels for easy draw and delivery of medication. The QD Syringe has its own low dead space mating needle Hub ~ www.QDSyringe.com